HUMAN TISSUE MAPPING CENTER (TMC)
The NIH has established a Cellular Senescence Network (SenNet) program, comprising of a national “tissue and cell mapping” effort involving 13 research institutes. Within the SenNet Consortium, the Buck Institute is a Tissue Mapping Center (TMC).
Buck Institute professors Judith Campisi, PhD and Birgit Schilling PhD, have been awarded a grant to participate in this study. The Buck Institute tissue mapping center (TMC) aims to characterize senescent cells -molecularly, spatially and during aging, in three very different human tissues (ovary, breast and skeletal muscle) and three biofluids (follicular fluid, plasma and urine). These samples have distinct biological characteristics and cell types that show significant changes with age. A separate grant awarded to Buck professor Simon Melov, PhD, will involve evaluating diverse technologies for detecting and validating senescent cells in ovarian tissue utilizing modern ‘organ on a chip’ models in collaboration with Francesca Duncan, PhD. The program as a whole, aims to unite cellular senescence researchers by providing publicly accessible atlases of senescent cells, the differences among them, and the molecules they release, using data collected from multiple human and model organism tissues.
Mapping senescence in the human body - a path to understanding aging
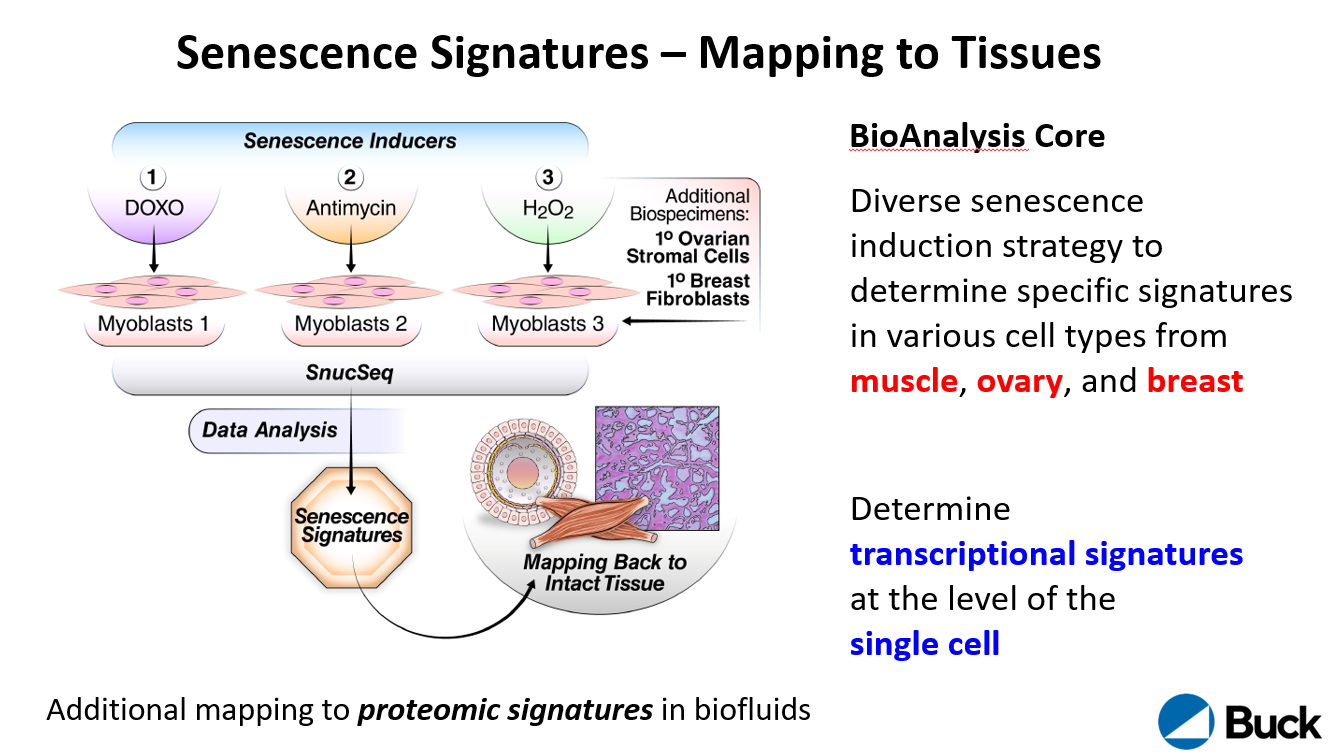
Cellular senescence is a multi-faceted cell fate that arrests cell proliferation and activates the synthesis and secretion of numerous cytokines, chemokines, growth factors, proteases and lipids, termed the Senescence Associated Secretory Phenotype (SASP). The SASP can influence tissue microenvironments, locally and distally, and thus senescent cells can strongly affect tissue function. Senescent cells (SnCs) increase with age in most vertebrate organisms, including mice and humans, and it is increasingly clear through both genetic and pharmacological manipulations that they can drive a growing list of age-related pathologies, ranging from neurodegeneration to cancer. At present, there are no invariant biomarkers of SnCs and the molecular characteristics of SnCs are remarkably heterogeneous and variable, depending on cell and tissue type, microenvironment, senescence inducer, and timing. Our overall goal is to determine, molecularly and spatially, when and where senescent cells occur in humans, and also how their patterns of gene expression and SASPs vary with tissue physiology and age. These goals are also the goals of the SenNet Consortium. We therefore propose to establish a SenNet tissue mapping center (TMC) comprised of an Administrative Core, Biospecimen Core, Biological Analysis Core and Data Analysis Core. Our proposed TMC will focus on three human tissues (ovary, breast and skeletal muscle) and three biofluids (follicular fluid, plasma and urine). These samples have distinct biological characteristics and cell types (somatic and reproductive; stromal, epithelial and vasculature) that show significant changes with age. All materials are available through established collaborations, subcontractors and/or biobanks (through the Biospecimen Core). Their analyses will include cutting-edge transcriptomic and proteomic techniques that will take advantage of the expertise of several Buck Institute investigators. Our preliminary data show that these tissues and biofluids are amenable to the state-of-the-art technologies proposed in the Biological Analysis and Data Analysis Cores, as well as new technologies proposed in our accompanying Technology Development application. Importantly, our findings will be applicable to many other human tissues and biofluids in order to encompass and complement the overall goals of the larger SenNet Consortium. The Administrative Core will coordinate all aspects of the TMC, from specimen acquisition to analysis, and will oversee and facilitate frequent interactions between the proposed TMC and other investigators, Cores and components of the SenNet Consortium.
Please visit NIH Senescence Network SenNet for more information.

CENTER DETAILS
-
 Birgit Schilling, PhD
Birgit Schilling, PhD -
 Malene Hansen, PhD
Malene Hansen, PhD -
 Pankaj Kapahi, PhD
Pankaj Kapahi, PhD -
 Simon Melov, PhD
Simon Melov, PhD -
 Eric Verdin, MD
Eric Verdin, MD -
 David Furman, PhD
David Furman, PhD -
 Francesca Duncan, PhD
Francesca Duncan, PhD -
 Chris Benz, PhD
Chris Benz, PhD -
 Akos Gerencser, PhD
Akos Gerencser, PhD
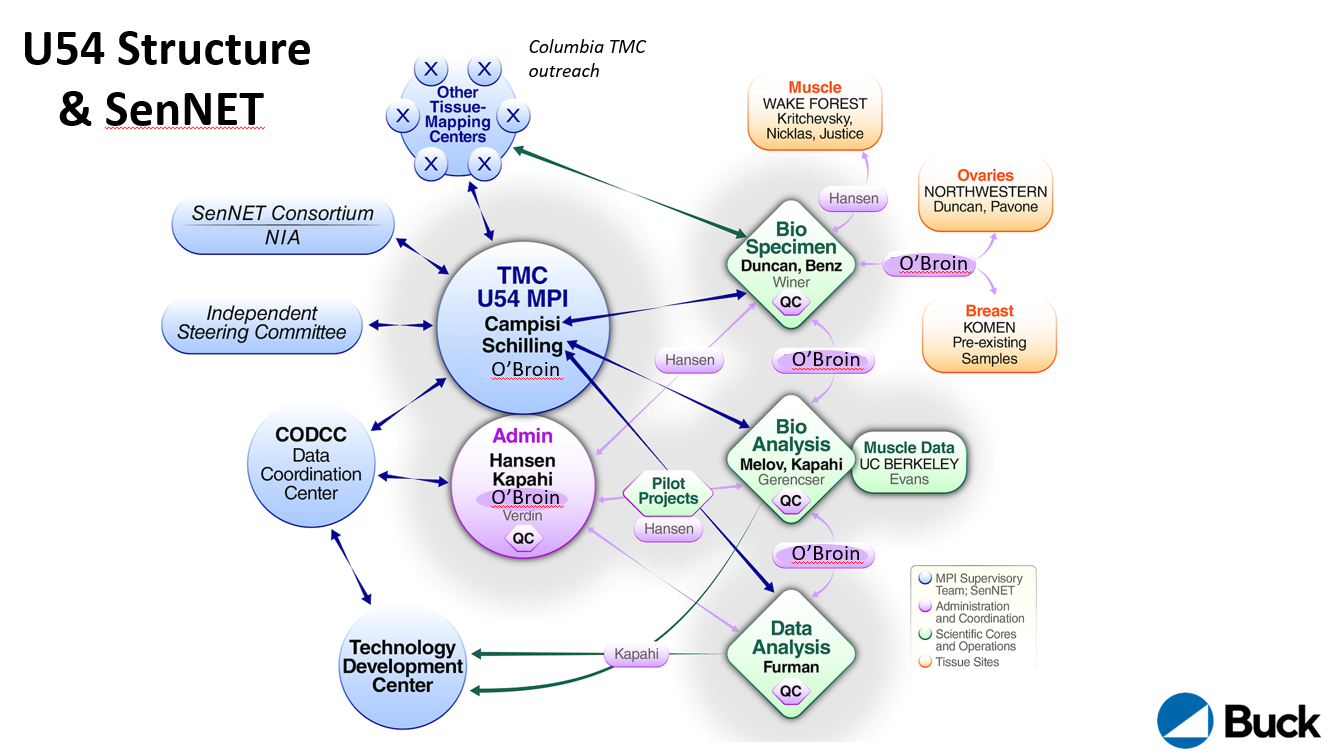
The Tissue Mapping Center (TMC) at the Buck Institute is led by Drs. Campisi and Schilling with Subcontracts to Wake Forest (Dr. Jamie Justice, muscle tissue collection), Northwestern University (Dr. Francesca Duncan, ovary tissue collection) and UC Berkeley (Dr. William Evans, muscle mass measurements).
The Admin Core is led by Drs. Malene Hansen, Pankaj Kapahi and Eric Verdin. The team oversees leadership plans, a tissue-mapping coordination plan and a network coordination plan, generating senescence tissue maps as part of the SenNet Consortium. It also ensures that data sharing pipelines within the TMC and with the SenNet network will be implemented and improved.
The Biospecimen Core is led by Drs. Francesca Duncan and Christopher Benz. The Biospecimen core partners with collaborators at Northwestern University, the Komen Tissue Bank, and Wake Forest University. The core procures, curates, validates, and distributes tissues to the Biological Analysis Core and other SenNet Tissue Mapping Centers.
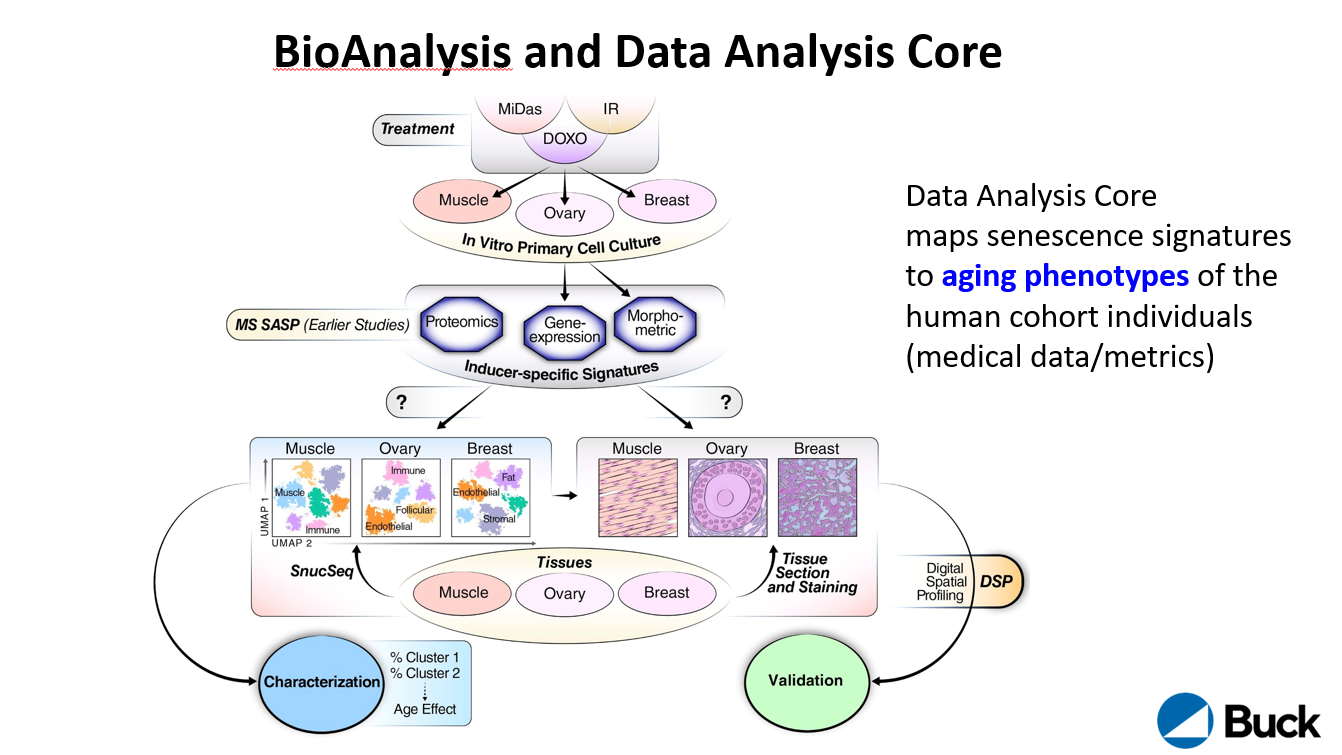
The Biological Analysis Core is led by Drs. Simon Melov, Pankaj Kapahi, Akos Gerencser, Birgit Schilling and Eric Verdin. The core will spatially map and determine the signatures of cellular senescence in healthy human ovaries, breast, and muscle in both sexes across an aging continuum.
The data analysis core is led by Dr. David Furman. The core aims to harness its ability to unbiasedly profile human tissues and blood to predict the senescent cell burden in humans and create an Atlas of SC biomarkers in tissues. The Data Analysis Core will provide pipelines for data processing, algorithms for data analysis, construct and share a map of human senescent cells in tissues.
Call for applications! Coming soon!
Malene Hansen
Admin Core Lead
Pankaj Kapahi
Admin Core Co-Lead
Mark Watson
U54 Coordinator
MWatson@buckinstitute.org
LAB GALLERY
Selected Publications
SenNet Consortium, NIH SenNet Consortium Mapping Senescent Cells in the Human Body to Understand Health and Disease. Nature Aging 2022 Dec; 2:1090–1100, https://doi.org/10.1038/s43587-022-00326-5
Kevin Perez, Serban Ciotlos,Julia McGirr, Chandani Limbad, Ryosuke Doi, Joshua P Nederveen, Mats I Nilsson, Daniel A Winer, William Evans, Mark Tarnopolsky, Judith Campisi, Simon Melov, Single nuclei profiling identifies cell specific markers of skeletal muscle aging, frailty, and senescence. PubMed volume 14, issue 23, pages 9393 – 9422 (2022), https://doi.org/10.18632/aging.204435