by Buck Institute
August 13, 2021 . BLOG
Old Dogs, New Tricks: New Roles for FGF21, a Well-Known Metabolic Molecule

 By Melia Granath-Panelo and Enrique Carrera, Dominican University of California Graduate Students
By Melia Granath-Panelo and Enrique Carrera, Dominican University of California Graduate Students
Edited by Dr. Pankaj Kapahi, Buck Professor
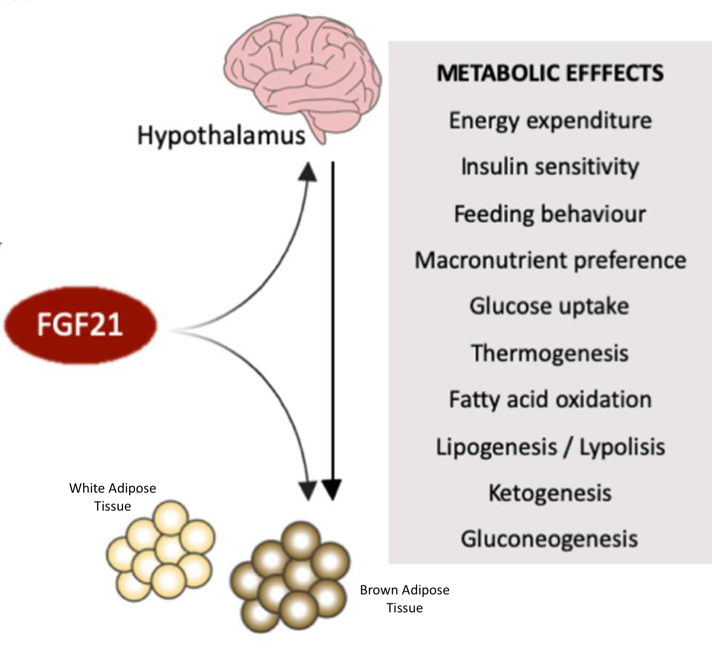
Human metabolism is a complex but beautiful system. Acting as an orchestra, our metabolism has many facets that come alive at specific times, which ultimately culminate in an intricate song that serves to return our bodies back to normalcy—a situation described by scientists as homeostasis. However, every good orchestra needs a conductor. Fibroblast Growth Factor-21, or FGF21, is a protein expressed in the liver and the fat tissue that controls many of the major drivers of our metabolism—glucose uptake, insulin sensitivity, blood lipid cholesterol levels, our body weight, and many others—appropriately earning the conductor title.
Furthermore, variations in the FGF21 gene have been implicated in the preference for sweet foods, providing a molecular mechanism for the “sweet tooth!” Although its definitive function is still being pieced together, FGF21 was thought to be secreted by the liver during fasting or ketogenic conditions, but the Mangelsdorf/Kliewer research team out of UT Southwestern Medical Center has brought a new mechanism onto the playing field.
Since diabetes is a metabolic disease, and FGF21 controls so many facets of our metabolism, do anti-diabetic medications depend on the actions of FGF21 to work? This was the fundamental question that motivated the research team. Using a class of anti-diabetic medications called thiazolidinediones, or TZDs for short, they found that not only did the presence of these compounds enhance FGF21 function, but also that TZDs depended on FGF21 to have any effect at all. TZDs lower insulin and glucose levels in the blood by stimulating a protein that controls glucose uptake in our cells, so this was a very interesting discovery in regards to the function of these molecules which are frequently prescribed to diabetes patients everywhere.
To further understand this mechanism, they generated a “knockout” mouse model that lacks FGF21 expression to study what happens when there is no FGF21 present, and treated both obese mice and the knockout mice with TZDs. Upon treatment, the team found that after feedingonly the obese mice were responsive to the beneficial actions of TZDs, showing improved glucose metabolism, improved insulin secretion, and lowered blood lipid levels. Because of these results, they concluded that FGF21 is necessary for treatment with TZDs to have any effect, and that treatment with TZDs can also cause FGF21 to be expressed after feeding in fat tissue.
Every new insight into these pathways brings us one step further to identifying potential treatments for diseases with metabolic implications, such as diabetes, cancer, and obesity. Not only does this study serve to prove a new mechanism for the actions of FGF21, it also showed a new way that a well-known therapeutic acts in the body. Although FGF21’s most prominent roles have been described above, emerging research suggests that FGF21 could have implications in the ketogenic diet, which has been shown to improve memory in aged mice and regulate our circadian clocks. (This comes from work done in the Verdin and Newman labs here at the Buck. Taken together, this work demonstrates how metabolism and aging often coalesce. There’s a reason why proper diet and adequate exercise are the mantra of aging scientists and physicians everywhere; what we put into our bodies governs how our bodies function from the system down to the molecular level! Proper consideration of these principles ensure that the conductors of our metabolism such as FGF21 can do their job—making sure that we can do our jobs in prime form and function.
Paul A. Dutchak, Takeshi Katafuchi, Angie L. Bookout, Jang Hyun Choi, Ruth T. Yu, David J. Mangelsdorf, and Steven A. Kliewer. (2012). Fibroblast Growth Factor-21 Regulates PPARγ Activity and the Antidiabetic Actions of Thiazolidinediones. Cell 148:556-567. DOI: 10.1016/j.cell.2011.11.062
SHARE