Gerencser Lab
The mitochondrial control of insulin secretion in type 2 diabetes
The life expectancy loss associated with T2D is average 6 years. A large fraction of this is associated with high blood glucose levels that is largely dependent on pancreatic β-cell function. Better understanding of basic mechanisms in β-cell function is expected to allow better nutraceutical or pharmacological support in T2D. In turn, this may significantly improve human healthspan through improving blood glucose control.
In non-diabetic individuals, insulin secretion can compensate for peripheral insulin resistance, while T2D is characterized by relative insulin insufficiency and a loss of compensation. Preventive or supportive therapies for T2D currently depend on the spontaneous recovery of β-cell function. Mitochondrial metabolism and bioenergetics are central to β-cell function and insulin secretion, and our goal is to identify how and why these change in T2D contributing to β-cell dysfunction.
We and others found an impaired metabolic response of the insulin-secreting pancreatic β-cells to glucose. This is hallmarked by a decreased response of mitochondria; a decreased glucose-stimulated hyperpolarization of the mitochondrial membrane potential (ΔψM)1. We identified a “mitochondrial signature of T2D” characteristic to β-cells from T2D individuals1, that we explain by an altered operation of an activation mechanism that controls mitochondrial metabolism2,3. Currently we study the mechanisms that control generation of ΔψM in diabetic and non-diabetic human pancreatic β-cells. We use live cell microscopy4,5, image analysis, and CRISPR-Cas9 and single-cell transcriptomics as primary tools to interrogate β-cell function and underlaying mechanisms.
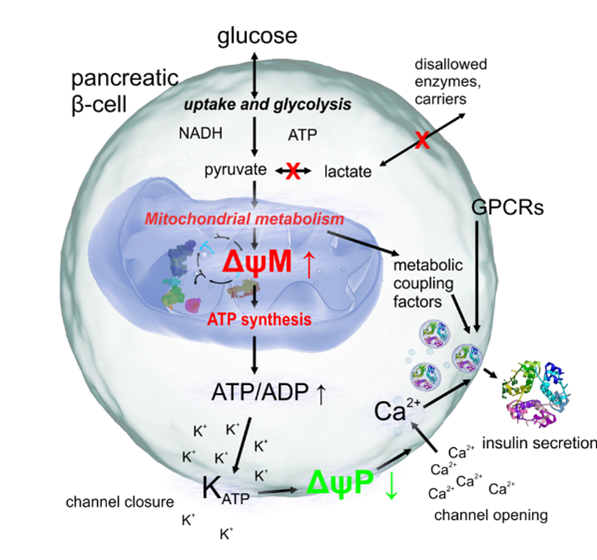
Pathways of insulin secretion
Mitochondria (blue) play a central “switch” role in the mechanism of glucose-stimulated insulin secretion in pancreatic β-cells. We found that this is an active amplification step3, rather than a simple linear pathway driven by nutrient availability. We investigate the mechanism of this activation step.
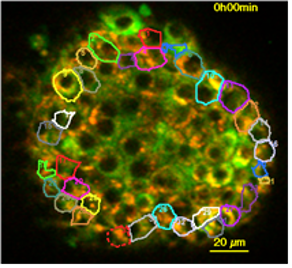
Human pancreatic islet with TMRM and FLIPR
Multiphoton microscopic imaging of live human islet stained with mitochondrial (red) and plasma membrane (green) potential probes. We use intact islet and dispersed islet cell imaging for functional assaying. We have developed some of the functional assays that we employ here.
Bibliography
- Gerencser, A. A. Bioenergetic Analysis of Single Pancreatic β-Cells Indicates an Impaired Metabolic Signature in Type 2 Diabetic Subjects. Endocrinology 156, 3496-3503 (2015). https://doi.org:10.1210/en.2015-1552
- Gerencser, A. A., Mookerjee, S. A., Jastroch, M. & Brand, M. D. Positive Feedback Amplifies the Response of Mitochondrial Membrane Potential to Glucose Concentration in Clonal Pancreatic Beta Cells. Biochimica et Biophysica Acta - Molecular Basis of Disease 1863, 1054-1065 (2017). https://doi.org:10.1016/j.bbadis.2016.10.015
- Gerencser, A. A. Metabolic activation-driven mitochondrial hyperpolarization predicts insulin secretion in human pancreatic beta-cells. Biochimica et Biophysica Acta (BBA) - Bioenergetics 1859, 817-828 (2018). https://doi.org:10.1016/j.bbabio.2018.06.006
- Gerencser, A. A., Mookerjee, S. A., Jastroch, M. & Brand, M. D. Measurement of the Absolute Magnitude and Time Courses of Mitochondrial Membrane Potential in Primary and Clonal Pancreatic Beta-Cells. PloS one 11, e0159199 (2016). https://doi.org:10.1371/journal.pone.0159199
- Lerner, C. A. & Gerencser, A. A. Unbiased Millivolts Assay of Mitochondrial Membrane Potential in Intact Cells. Methods Mol Biol 2497, 11-61 (2022). https://doi.org:10.1007/978-1-0716-2309-1_2
Science is showing that while chronological aging is inevitable, biological aging is malleable. There's a part of it that you can fight, and we are getting closer and closer to winning that fight.
Eric Verdin, MD, Buck Institute President and CEO